Sodium Hydroxide Density G Ml
 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sodium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Appearance | silvery white metallic | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard diminutive weight A r°(Na) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sodium in the periodic table | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic number (Z) | xi | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Group | group i: hydrogen and brine metals | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Catamenia | catamenia 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cake | due south-cake | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Ne] 3si | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, ane | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concrete properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase atSTP | solid | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting indicate | 370.944 Chiliad (97.794 °C, 208.029 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 1156.090 One thousand (882.940 °C, 1621.292 °F) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (nearr.t.) | 0.968 g/cm3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| when liquid (atk.p.) | 0.927 g/cmthree | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Critical point | 2573 K, 35 MPa (extrapolated) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 2.60 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rut of vaporization | 97.42 kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 28.230 J/(mol·K) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | −1, 0,[two] +one (a strongly basic oxide) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 0.93 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | empirical: 186 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 166±9 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Van der Waals radius | 227 pm | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other backdrop | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Natural occurrence | primordial | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | torso-centered cubic (bcc) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound thin rod | 3200 m/south (at 20 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | 71 µm/(m⋅Grand) (at 25 °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal electrical conductivity | 142 W/(m⋅G) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Electric resistivity | 47.7 nΩ⋅m (at twenty °C) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | paramagnetic[iii] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar magnetic susceptibility | +16.0×10−6 cmthree/mol (298 K)[4] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immature's modulus | ten GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 3.iii GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 6.3 GPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 0.5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 0.69 MPa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Number | 7440-23-5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery and first isolation | Humphry Davy (1807) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symbol | "Na": from New Latin natrium, coined from German Natron, 'natron' | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Main isotopes of sodium | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Sodium is a chemical chemical element with the symbolNa (from Latin natrium) and atomic number 11. It is a soft, argent-white, highly reactive metal. Sodium is an brine metal, being in group 1 of the periodic table. Its just stable isotope is 23Na. The gratis metal does non occur in nature, and must be prepared from compounds. Sodium is the sixth most abundant chemical element in the Earth's crust and exists in numerous minerals such every bit feldspars, sodalite, and halite (NaCl). Many salts of sodium are highly water-soluble: sodium ions accept been leached by the activeness of water from the Earth's minerals over eons, and thus sodium and chlorine are the nearly common dissolved elements by weight in the oceans.
Sodium was first isolated by Humphry Davy in 1807 by the electrolysis of sodium hydroxide. Among many other useful sodium compounds, sodium hydroxide (lye) is used in soap manufacture, and sodium chloride (edible salt) is a de-icing agent and a food for animals including humans.
Sodium is an essential element for all animals and some plants. Sodium ions are the major cation in the extracellular fluid (ECF) and as such are the major contributor to the ECF osmotic pressure and ECF compartment volume.[ commendation needed ] Loss of h2o from the ECF compartment increases the sodium concentration, a condition called hypernatremia. Isotonic loss of h2o and sodium from the ECF compartment decreases the size of that compartment in a status chosen ECF hypovolemia.
By ways of the sodium-potassium pump, living human cells pump three sodium ions out of the cell in exchange for two potassium ions pumped in; comparing ion concentrations across the cell membrane, inside to outside, potassium measures nigh 40:1, and sodium, most one:10. In nerve cells, the electric charge across the jail cell membrane enables transmission of the nerve impulse—an action potential—when the charge is dissipated; sodium plays a fundamental role in that activity.
Characteristics
Physical

Sodium at standard temperature and force per unit area is a soft silvery metallic that combines with oxygen in the air and forms grayish white sodium oxide unless immersed in oil or inert gas, which are the conditions it is usually stored in. Sodium metal can be easily cut with a pocketknife and is a adept conductor of electricity and estrus because it has simply one electron in its valence shell, resulting in weak metal bonding and gratuitous electrons, which comport energy. Due to having low atomic mass and big diminutive radius, sodium is third-to the lowest degree dense of all elemental metals and is one of simply 3 metals that tin can bladder on water, the other two beingness lithium and potassium.[5]
The melting (98 °C) and boiling (883 °C) points of sodium are lower than those of lithium but higher than those of the heavier alkali metals potassium, rubidium, and caesium, following periodic trends downward the grouping.[6] These properties modify dramatically at elevated pressures: at 1.5 Mbar, the color changes from silvery metallic to black; at 1.9 Mbar the fabric becomes transparent with a ruby-red color; and at three Mbar, sodium is a clear and transparent solid. All of these loftier-pressure allotropes are insulators and electrides.[7]

A positive flame test for sodium has a bright xanthous colour.
In a flame test, sodium and its compounds glow yellowish[8] because the excited 3s electrons of sodium emit a photon when they fall from 3p to 3s; the wavelength of this photon corresponds to the D line at about 589.3 nm. Spin-orbit interactions involving the electron in the 3p orbital divide the D line into two, at 589.0 and 589.6 nm; hyperfine structures involving both orbitals cause many more lines.[nine]
Isotopes
Twenty isotopes of sodium are known, but only 23Na is stable. 23Na is created in the carbon-burning procedure in stars by fusing two carbon atoms together; this requires temperatures above 600 megakelvins and a star of at least 3 solar masses.[10] Two radioactive, cosmogenic isotopes are the byproduct of cosmic ray spallation: 22Na has a one-half-life of 2.6 years and 24Na, a half-life of 15 hours; all other isotopes have a half-life of less than i minute.[eleven]
Two nuclear isomers have been discovered, the longer-lived ane beingness 24mNa with a half-life of effectually 20.2 milliseconds. Acute neutron radiation, as from a nuclear criticality accident, converts some of the stable 23Na in human claret to 24Na; the neutron radiation dosage of a victim can be calculated by measuring the concentration of 24Na relative to 23Na.[12]
Chemistry
Sodium atoms have eleven electrons, ane more than the stable configuration of the noble gas neon. The start and second ionization energies are 495.8 kJ/mol and 4562 kJ/mol, respectively. As a result, sodium usually forms ionic compounds involving the Na+ cation.[13]
Metallic sodium
Metallic sodium is generally less reactive than potassium and more than reactive than lithium.[14] Sodium metal is highly reducing, with the standard reduction potential for the Na+/Na couple existence −two.71 volts,[15] though potassium and lithium have even more negative potentials.[16] The thermal, fluidic, chemical, and nuclear backdrop of molten sodium metal have caused it to be one of the main coolants of choice for the fast breeder reactor. Such nuclear reactors are seen as a crucial step for the production of clean energy.[17]
Salts and oxides

The structure of sodium chloride, showing octahedral coordination around Na+ and Cl− centres. This framework disintegrates when dissolved in water and reassembles when the water evaporates.
Sodium compounds are of immense commercial importance, existence especially central to industries producing glass, paper, soap, and textiles.[eighteen] The most of import sodium compounds are table table salt (NaCl), soda ash (NatwoCOiii), baking soda (NaHCO3), caustic soda (NaOH), sodium nitrate (NaNO3), di- and tri-sodium phosphates, sodium thiosulfate (NaiiS2Othree·5HtwoO), and borax (Na2B4O7·10HiiO).[nineteen] In compounds, sodium is usually ionically bonded to water and anions and is viewed as a hard Lewis acid.[xx]

Two equivalent images of the chemical structure of sodium stearate, a typical soap.
Most soaps are sodium salts of fatty acids. Sodium soaps have a higher melting temperature (and seem "harder") than potassium soaps.[19] Sodium containing mixed oxides are promising catalysts[21] and photocatalysts.[22] Photochemically intercalated sodium ion enhances the photoelectrocatalytic activity of WO3.[23]
Similar all the alkali metals, sodium reacts exothermically with water. The reaction produces caustic soda (sodium hydroxide) and flammable hydrogen gas. When burned in air, information technology forms primarily sodium peroxide with some sodium oxide.[24]
Aqueous solutions
Sodium tends to class water-soluble compounds, such as halides, sulfates, nitrates, carboxylates and carbonates. The main aqueous species are the aquo complexes [Na(H2O) n ]+, where northward = 4–8; with n = 6 indicated from X-ray diffraction data and computer simulations.[25]
Direct precipitation of sodium salts from aqueous solutions is rare because sodium salts typically accept a high affinity for h2o. An exception is sodium bismuthate (NaBiOthree).[26] Because of the high solubility of its compounds, sodium salts are usually isolated equally solids by evaporation or by atmospheric precipitation with an organic antisolvent, such as ethanol; for case, only 0.35 g/Fifty of sodium chloride volition dissolve in ethanol.[27] Crown ethers, like fifteen-crown-v, may be used equally a stage-transfer goad.[28]
Sodium content of samples is determined by atomic assimilation spectrophotometry or past potentiometry using ion-selective electrodes.[29]
Electrides and sodides
Like the other alkali metals, sodium dissolves in ammonia and some amines to give deeply colored solutions; evaporation of these solutions leaves a shiny motion-picture show of metallic sodium. The solutions contain the coordination complex (Na(NH3)6)+, with the positive charge balanced by electrons as anions; cryptands permit the isolation of these complexes as crystalline solids. Sodium forms complexes with crown ethers, cryptands and other ligands.[thirty]
For instance, fifteen-crown-5 has a high affinity for sodium because the cavity size of 15-crown-five is 1.7–ii.two Å, which is plenty to fit the sodium ion (one.nine Å).[31] [32] Cryptands, similar crown ethers and other ionophores, also have a high analogousness for the sodium ion; derivatives of the alkalide Na− are obtainable[33] by the add-on of cryptands to solutions of sodium in ammonia via disproportionation.[34]
Organosodium compounds

The construction of the circuitous of sodium (Na+, shown in yellowish) and the antibody monensin-A.
Many organosodium compounds have been prepared. Because of the high polarity of the C-Na bonds, they behave like sources of carbanions (salts with organic anions). Some well-known derivatives include sodium cyclopentadienide (NaC5H5) and trityl sodium ((C6H5)iiiCNa).[35] Sodium naphthalene, Na+[C10H8•]−, a strong reducing agent, forms upon mixing Na and naphthalene in ethereal solutions.[36]
Intermetallic compounds
Sodium forms alloys with many metals, such as potassium, calcium, lead, and the grouping 11 and 12 elements. Sodium and potassium class KNa2 and NaK. NaK is xl–90% potassium and information technology is liquid at ambient temperature. It is an excellent thermal and electrical usher. Sodium-calcium alloys are by-products of the electrolytic production of sodium from a binary common salt mixture of NaCl-CaCl2 and ternary mixture NaCl-CaCl2-BaCl2. Calcium is but partially miscible with sodium, and the ane-two% of information technology dissolved in the sodium obtained from said mixtures can be precipitated by cooling to 120 °C and filtering.[37]
In a liquid state, sodium is completely miscible with lead. There are several methods to make sodium-atomic number 82 alloys. 1 is to melt them together and another is to eolith sodium electrolytically on molten atomic number 82 cathodes. NaPbiii, NaPb, Na9Pb4, Na5Pb2, and NaxvLead4 are some of the known sodium-pb alloys. Sodium as well forms alloys with gold (NaAu2) and silvery (NaAg2). Group 12 metals (zinc, cadmium and mercury) are known to make alloys with sodium. NaZn13 and NaCd2 are alloys of zinc and cadmium. Sodium and mercury course NaHg, NaHg4, NaHg2, Na3Hg2, and NathreeHg.[38]
History
Because of its importance in human health, common salt has long been an of import commodity, as shown past the English word salary, which derives from salarium, the wafers of table salt sometimes given to Roman soldiers forth with their other wages. In medieval Europe, a chemical compound of sodium with the Latin name of sodanum was used as a headache remedy. The name sodium is idea to originate from the Arabic suda, pregnant headache, equally the headache-alleviating properties of sodium carbonate or soda were well known in early times.[39]
Although sodium, sometimes called soda, had long been recognized in compounds, the metallic itself was not isolated until 1807 by Sir Humphry Davy through the electrolysis of sodium hydroxide.[40] [41] In 1809, the German physicist and chemist Ludwig Wilhelm Gilbert proposed the names Natronium for Humphry Davy'south "sodium" and Kalium for Davy'due south "potassium".[42]
The chemical abridgement for sodium was first published in 1814 by Jöns Jakob Berzelius in his arrangement of atomic symbols,[43] [44] and is an abbreviation of the element's New Latin name natrium, which refers to the Egyptian natron,[39] a natural mineral salt mainly consisting of hydrated sodium carbonate. Natron historically had several important industrial and household uses, later on eclipsed by other sodium compounds.[45]
Sodium imparts an intense yellow color to flames. As early every bit 1860, Kirchhoff and Bunsen noted the high sensitivity of a sodium flame examination, and stated in Annalen der Physik und Chemie:[46]
In a corner of our threescore grand3 room farthest away from the apparatus, we exploded 3 mg of sodium chlorate with milk saccharide while observing the nonluminous flame before the slit. After a while, it glowed a vivid yellowish and showed a stiff sodium line that disappeared only after 10 minutes. From the weight of the sodium salt and the volume of air in the room, we easily calculate that ane part by weight of air could not contain more than 1/20 millionth weight of sodium.
Occurrence
The Earth's crust contains two.27% sodium, making it the 7th most abundant chemical element on Earth and the fifth almost abundant metal, behind aluminium, iron, calcium, and magnesium and ahead of potassium.[47] Sodium'south estimated oceanic abundance is 10.eight grams per liter.[48] Because of its high reactivity, it is never plant as a pure element. It is found in many minerals, some very soluble, such every bit halite and natron, others much less soluble, such as amphibole and zeolite. The insolubility of sure sodium minerals such as cryolite and feldspar arises from their polymeric anions, which in the case of feldspar is a polysilicate.
Astronomical observations
Atomic sodium has a very potent spectral line in the xanthous-orange part of the spectrum (the same line equally is used in sodium-vapour street lights). This appears every bit an absorption line in many types of stars, including the Sun. The line was start studied in 1814 past Joseph von Fraunhofer during his investigation of the lines in the solar spectrum, now known as the Fraunhofer lines. Fraunhofer named it the "D" line, although information technology is now known to actually be a group of closely spaced lines split past a fine and hyperfine structure.[49]
The force of the D line allows its detection in many other astronomical environments. In stars, information technology is seen in any whose surfaces are cool enough for sodium to exist in diminutive form (rather than ionised). This corresponds to stars of roughly F-type and cooler. Many other stars appear to have a sodium assimilation line, but this is really caused by gas in the foreground interstellar medium. The two can be distinguished via high-resolution spectroscopy, because interstellar lines are much narrower than those broadened by stellar rotation.[50]
Sodium has also been detected in numerous Solar System environments, including Mercury's atmosphere,[51] the exosphere of the Moon,[52] and numerous other bodies. Some comets accept a sodium tail,[53] which was first detected in observations of Comet Hale–Bopp in 1997.[54] Sodium has even been detected in the atmospheres of some extrasolar planets via transit spectroscopy.[55]
Commercial production
Employed but in rather specialized applications, but near 100,000 tonnes of metallic sodium are produced annually.[18] Metallic sodium was first produced commercially in the late 19th century[37] by carbothermal reduction of sodium carbonate at 1100 °C, as the first step of the Deville process for the production of aluminium:[56] [57] [58]
- NatwoCO3 + 2 C → 2 Na + 3 CO
The loftier need for aluminium created the need for the production of sodium. The introduction of the Hall–Héroult process for the production of aluminium by electrolysing a molten common salt bath ended the need for large quantities of sodium. A related process based on the reduction of sodium hydroxide was developed in 1886.[56]
Sodium is at present produced commercially through the electrolysis of molten sodium chloride, based on a process patented in 1924.[59] [threescore] This is done in a Downs jail cell in which the NaCl is mixed with calcium chloride to lower the melting betoken below 700 °C.[61] Equally calcium is less electropositive than sodium, no calcium will exist deposited at the cathode.[62] This method is less expensive than the previous Castner process (the electrolysis of sodium hydroxide).[63] If sodium of high purity is required, it tin exist distilled once or several times.
The marketplace for sodium is volatile due to the difficulty in its storage and shipping; information technology must be stored under a dry inert gas temper or anhydrous mineral oil to prevent the formation of a surface layer of sodium oxide or sodium superoxide.[64]
Uses
Though metallic sodium has some of import uses, the major applications for sodium utilise compounds; millions of tons of sodium chloride, hydroxide, and carbonate are produced annually. Sodium chloride is extensively used for anti-icing and de-icing and as a preservative; examples of the uses of sodium bicarbonate include baking, as a raising amanuensis, and sodablasting. Forth with potassium, many important medicines have sodium added to ameliorate their bioavailability; though potassium is the better ion in most cases, sodium is chosen for its lower price and diminutive weight.[65] Sodium hydride is used every bit a base of operations for various reactions (such every bit the aldol reaction) in organic chemistry,[66] and every bit a reducing agent in inorganic chemistry.[67]
Metallic sodium is used mainly for the production of sodium borohydride, sodium azide, indigo, and triphenylphosphine. A once-common use was the making of tetraethyllead and titanium metal; because of the motion away from TEL and new titanium production methods, the production of sodium declined after 1970.[18] Sodium is also used every bit an alloying metal, an anti-scaling agent,[68] and as a reducing agent for metals when other materials are ineffective.
Note the gratis element is non used every bit a scaling agent, ions in the water are exchanged for sodium ions. Sodium plasma ("vapor") lamps are often used for street lighting in cities, shedding light that ranges from yellow-orangish to peach every bit the force per unit area increases.[69] By itself or with potassium, sodium is a desiccant; it gives an intense blue coloration with benzophenone when the desiccate is dry.[70]
In organic synthesis, sodium is used in diverse reactions such as the Birch reduction, and the sodium fusion test is conducted to qualitatively analyse compounds.[71] Sodium reacts with alcohol and gives alkoxides, and when sodium is dissolved in ammonia solution, information technology can be used to reduce alkynes to trans-alkenes.[72] [73] Lasers emitting light at the sodium D line are used to create artificial laser guide stars that assist in the adaptive optics for land-based visible-low-cal telescopes.[74]
Heat transfer
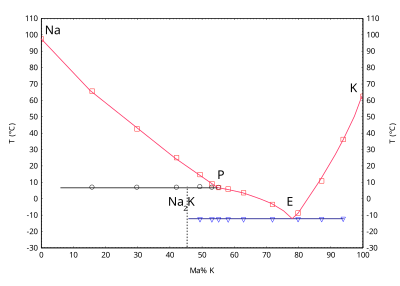
NaK stage diagram, showing the melting indicate of sodium as a part of potassium concentration. NaK with 77% potassium is eutectic and has the lowest melting signal of the NaK alloys at −12.6 °C.[75]
Liquid sodium is used as a heat transfer fluid in sodium-cooled fast reactors[76] because it has the high thermal conductivity and low neutron absorption cross section required to attain a high neutron flux in the reactor.[77] The high boiling point of sodium allows the reactor to operate at ambient (normal) pressure,[77] but drawbacks include its opacity, which hinders visual maintenance, and its strongly reducing properties. Sodium will explode in contact with water, although it will but burn gently in air. [78]
Radioactive sodium-24 may be produced past neutron bombardment during operation, posing a slight radiations run a risk; the radioactivity stops inside a few days after removal from the reactor.[79] If a reactor needs to exist close down frequently, NaK is used. Because NaK is a liquid at room temperature, the coolant does not solidify in the pipes.[lxxx]
In this case, the pyrophoricity of potassium requires extra precautions to prevent and find leaks.[81] Another heat transfer awarding is poppet valves in high-functioning internal combustion engines; the valve stems are partially filled with sodium and work as a heat piping to cool the valves.[82]
Biological part
Biological role in humans
In humans, sodium is an essential mineral that regulates claret volume, blood force per unit area, osmotic equilibrium and pH. The minimum physiological requirement for sodium is estimated to range from about 120 milligrams per mean solar day in newborns to 500 milligrams per day over the historic period of 10.[83]
Nutrition
Sodium chloride is the principal source of sodium in the nutrition, and is used as seasoning and preservative in such commodities as pickled preserves and jerky; for Americans, most sodium chloride comes from processed foods.[84] Other sources of sodium are its natural occurrence in food and such food additives every bit monosodium glutamate (MSG), sodium nitrite, sodium saccharin, baking soda (sodium bicarbonate), and sodium benzoate.[85]
The U.Due south. Found of Medicine prepare its tolerable upper intake level for sodium at 2.3 grams per day,[86] but the average person in the United States consumes three.4 grams per solar day.[87] The American Heart Association recommends no more than i.5 g of sodium per solar day.[88]
High sodium consumption
High sodium consumption is unhealthy, and can atomic number 82 to alteration in the mechanical functioning of the heart.[89] High sodium consumption is also associated with chronic kidney disease, loftier blood pressure, cardiovascular diseases, and stroke.[89]
High blood pressure
There is a strong correlation betwixt higher sodium intake and higher blood pressure.[xc] Studies have found that lowering sodium intake by two thou per mean solar day tends to lower systolic claret force per unit area past about 2 to four mm Hg.[91] It has been estimated that such a decrease in sodium intake would lead to between ix and 17% fewer cases of hypertension.[91]
Hypertension causes 7.6 1000000 premature deaths worldwide each yr.[92] (Annotation that table salt contains about 39.iii% sodium[93]—the rest being chlorine and trace chemicals; thus, 2.3 g sodium is almost 5.9 g, or 5.iii ml, of table salt—well-nigh ane Usa teaspoon.[94] [95])
One study constitute that people with or without hypertension who excreted less than 3 grams of sodium per day in their urine (and therefore were taking in less than 3 g/d) had a college risk of death, stroke, or heart set on than those excreting four to 5 grams per day.[96] Levels of seven g per day or more than in people with hypertension were associated with higher mortality and cardiovascular events, but this was not found to be true for people without hypertension.[96] The Usa FDA states that adults with hypertension and prehypertension should reduce daily sodium intake to 1.5 g.[95]
Physiology
The renin–angiotensin system regulates the amount of fluid and sodium concentration in the body. Reduction of claret force per unit area and sodium concentration in the kidney outcome in the product of renin, which in turn produces aldosterone and angiotensin, which stimulates the reabsorption of sodium back into the bloodstream. When the concentration of sodium increases, the production of renin decreases, and the sodium concentration returns to normal.[97] The sodium ion (Na+) is an important electrolyte in neuron function, and in osmoregulation between cells and the extracellular fluid. This is accomplished in all animals by Na+/K+-ATPase, an agile transporter pumping ions against the gradient, and sodium/potassium channels.[98] Sodium is the about prevalent metal ion in extracellular fluid.[99]
In humans, unusually depression or high sodium levels in the claret is recognized in medicine as hyponatremia and hypernatremia. These conditions may be caused by genetic factors, ageing, or prolonged airsickness or diarrhea.[100]
Biological function in plants
In C4 plants, sodium is a micronutrient that aids metabolism, specifically in regeneration of phosphoenolpyruvate and synthesis of chlorophyll.[101] In others, information technology substitutes for potassium in several roles, such as maintaining turgor pressure and aiding in the opening and closing of stomata.[102] Backlog sodium in the soil tin limit the uptake of water by decreasing the water potential, which may result in plant wilting; excess concentrations in the cytoplasm can lead to enzyme inhibition, which in turn causes necrosis and chlorosis.[103]
In response, some plants have adult mechanisms to limit sodium uptake in the roots, to store it in cell vacuoles, and restrict salt transport from roots to leaves.[104] Excess sodium may also be stored in sometime establish tissue, limiting the harm to new growth. Halophytes have adapted to be able to flourish in sodium rich environments.[104]
Rubber and precautions
| Hazards | |
|---|---|
| GHS labelling: | |
| Pictograms |   |
| Signal give-and-take | Danger |
| Hazard statements | H260, H314 |
| Precautionary statements | P223, P231+P232, P280, P305+P351+P338, P370+P378, P422 [105] |
| NFPA 704 (burn diamond) | [106] 3 two 2 |
Sodium forms flammable hydrogen and caustic sodium hydroxide on contact with water;[107] ingestion and contact with moisture on pare, eyes or mucous membranes tin can cause severe burns.[108] [109] Sodium spontaneously explodes in the presence of water due to the formation of hydrogen (highly explosive) and sodium hydroxide (which dissolves in the h2o, liberating more than surface). However, sodium exposed to air and ignited or reaching autoignition (reported to occur when a molten pool of sodium reaches most 290 °C, 554 °F)[110] displays a relatively mild burn.
In the case of massive (non-molten) pieces of sodium, the reaction with oxygen eventually becomes ho-hum due to formation of a protective layer.[111] Burn extinguishers based on h2o accelerate sodium fires. Those based on carbon dioxide and bromochlorodifluoromethane should not be used on sodium fire.[109] Metal fires are Class D, just not all Course D extinguishers are constructive when used to extinguish sodium fires. An constructive extinguishing agent for sodium fires is Met-L-X.[109] Other effective agents include Lith-X, which has graphite powder and an organophosphate flame retardant, and dry out sand.[112]
Sodium fires are prevented in nuclear reactors by isolating sodium from oxygen with surrounding pipes containing inert gas.[113] Puddle-type sodium fires are prevented using diverse pattern measures called grab pan systems. They collect leaking sodium into a leak-recovery tank where information technology is isolated from oxygen.[113]
Liquid sodium fires are more than dangerous to handle than solid sodium fires, especially if in that location is insufficient experience with the safe handling of molten sodium. In a technical report for the United States Burn Assistants,[114] R. J. Gordon writes (emphasis in original)
Once ignited, sodium is very hard to extinguish. It will react violently with h2o, as noted previously, and with any extinguishing agent that contains water. It volition also react with many other common extinguishing agents, including carbon dioxide and the halogen compounds and well-nigh dry chemical agents. The but rubber and constructive extinguishing agents are completely dry out inert materials, such as Grade D extinguishing agents, soda ash, graphite, diatomaceous earth, or sodium chloride, all of which can be used to coffin a pocket-size quantity of burning sodium and exclude oxygen from reaching the metallic.
The extinguishing agent must be absolutely dry, every bit even a trace of water in the fabric can react with the called-for sodium to cause an explosion. Sodium chloride is recognized as an extinguishing medium because of its chemical stability, however it is hydroscopic (has the property of alluring and holding water molecules on the surface of the salt crystals) and must exist kept admittedly dry to be used safely as an extinguishing amanuensis. Every crystal of sodium chloride likewise contains a trace quantity of moisture within the structure of the crystal.
Molten sodium is extremely dangerous because it is much more reactive than a solid mass. In the liquid grade, every sodium atom is gratuitous and mobile to instantaneously combine with any available oxygen atom or other oxidizer, and any gaseous by-product will be created as a speedily expanding gas bubble within the molten mass. Even a infinitesimal amount of water tin can create this type of reaction. Any corporeality of water introduced into a puddle of molten sodium is probable to cause a trigger-happy explosion within the liquid mass, releasing the hydrogen as a rapidly expanding gas and causing the molten sodium to erupt from the container.
When molten sodium is involved in a fire, the combustion occurs at the surface of the liquid. An inert gas, such as nitrogen or argon, can be used to course an inert layer over the puddle of burning liquid sodium, but the gas must be applied very gently and independent over the surface. Except for soda ash, most of the powdered agents that are used to extinguish minor fires in solid pieces or shallow pools will sink to the bottom of a molten mass of burning sodium -- the sodium will bladder to the meridian and continue to burn. If the burning sodium is in a container, it may be feasible to extinguish the burn down past placing a lid on the container to exclude oxygen.
See also
References
- ^ "Standard Atomic Weights: Sodium". CIAAW. 2005.
- ^ The compound NaCl has been shown in experiments to exists in several unusual stoichiometries under high pressure, including NathreeCl in which contains a layer of sodium(0) atoms; run into Zhang, W.; Oganov, A. R.; Goncharov, A. F.; Zhu, Q.; Boulfelfel, Southward. E.; Lyakhov, A. O.; Stavrou, E.; Somayazulu, M.; Prakapenka, V. B.; Konôpková, Z. (2013). "Unexpected Stable Stoichiometries of Sodium Chlorides". Science. 342 (6165): 1502–1505. arXiv:1310.7674. Bibcode:2013Sci...342.1502Z. doi:10.1126/science.1244989. PMID 24357316. S2CID 15298372.
- ^ Magnetic susceptibility of the elements and inorganic compounds, in Lide, D. R., ed. (2005). CRC Handbook of Chemical science and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN0-8493-0486-5.
- ^ Weast, Robert (1984). CRC, Handbook of Chemistry and Physics. Boca Raton, Florida: Chemical Rubber Company Publishing. pp. E110. ISBN0-8493-0464-iv.
- ^ Greenwood and Earnshaw, p. 75
- ^ ""Alkali Metals." Science of Everyday Things". Encyclopedia.com. Archived from the original on 17 October 2016. Retrieved fifteen October 2016.
- ^ Gatti, M.; Tokatly, I.; Rubio, A. (2010). "Sodium: A Charge-Transfer Insulator at High Pressures". Physical Review Messages. 104 (21): 216404. arXiv:1003.0540. Bibcode:2010PhRvL.104u6404G. doi:10.1103/PhysRevLett.104.216404. PMID 20867123. S2CID 18359072.
- ^ Schumann, Walter (5 August 2008). Minerals of the World (second ed.). Sterling. p. 28. ISBN978-1-4027-5339-8. OCLC 637302667.
- ^ Citron, Yard. 50.; Gabel, C.; Stroud, C.; Stroud, C. (1977). "Experimental Study of Power Broadening in a Ii-Level Atom". Physical Review A. 16 (4): 1507–1512. Bibcode:1977PhRvA..sixteen.1507C. doi:ten.1103/PhysRevA.16.1507.
- ^ Denisenkov, P. A.; Ivanov, V. V. (1987). "Sodium Synthesis in Hydrogen Called-for Stars". Soviet Astronomy Letters. 13: 214. Bibcode:1987SvAL...13..214D.
- ^ Audi, Georges; Bersillon, Olivier; Blachot, Jean; Wapstra, Aaldert Hendrik (2003), "The NorthwardUBASE evaluation of nuclear and decay backdrop", Nuclear Physics A, 729: 3–128, Bibcode:2003NuPhA.729....3A, doi:10.1016/j.nuclphysa.2003.11.001
- ^ Sanders, F. Due west.; Auxier, J. A. (1962). "Neutron Activation of Sodium in Anthropomorphous Phantoms". Health Physics. viii (4): 371–379. doi:x.1097/00004032-196208000-00005. PMID 14496815. S2CID 38195963.
- ^ Lawrie Ryan; Roger Norris (31 July 2014). Cambridge International Every bit and A Level Chemistry Coursebook (illustrated ed.). Cambridge University Press, 2014. p. 36. ISBN978-1-107-63845-7.
- ^ De Leon, N. "Reactivity of Alkali Metals". Indiana University Northwest. Archived from the original on 16 October 2018. Retrieved vii December 2007.
- ^ Atkins, Peter W.; de Paula, Julio (2002). Physical Chemical science (7th ed.). Westward. H. Freeman. ISBN978-0-7167-3539-7. OCLC 3345182.
- ^ Davies, Julian A. (1996). Synthetic Coordination Chemistry: Principles and Practice. World Scientific. p. 293. ISBN978-981-02-2084-half dozen. OCLC 717012347.
- ^ "Fast Neutron Reactors | FBR - World Nuclear Association". World-nuclear.org. Retrieved 4 October 2022.
- ^ a b c Alfred Klemm, Gabriele Hartmann, Ludwig Lange, "Sodium and Sodium Alloys" in Ullmann's Encyclopedia of Industrial Chemistry 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a24_277
- ^ a b Holleman, Arnold F.; Wiberg, Egon; Wiberg, Nils (1985). Lehrbuch der Anorganischen Chemie (in German) (91–100 ed.). Walter de Gruyter. pp. 931–943. ISBN978-3-11-007511-iii.
- ^ Cowan, James A. (1997). Inorganic Biochemistry: An Introduction. Wiley-VCH. p. 7. ISBN978-0-471-18895-7. OCLC 34515430.
- ^ Kim, Heeyeon; Lee, Suhyeong; Jang, Seoyoung; Yu, Ji-haeng; Yoo, Jong Suk; Oh, Jangwon (5 September 2021). "Outcome of facile nitrogen doping on catalytic performance of NaW/Mn/SiO2 for oxidative coupling of marsh gas". Applied Catalysis B: Environmental. 292: 120161. doi:10.1016/j.apcatb.2021.120161. ISSN 0926-3373. Archived from the original on viii January 2022. Retrieved eight Jan 2022.
- ^ Praxedes, Fabiano R.; Nobre, Marcos A. L.; Poon, Po S.; Matos, Juan; Lanfredi, Silvania (five Dec 2021). "Nanostructured KxNa1-xNbO3 hollow spheres equally potential materials for the photocatalytic treatment of polluted h2o". Applied Catalysis B: Environmental. 298: 120502. doi:10.1016/j.apcatb.2021.120502. ISSN 0926-3373. Archived from the original on 8 January 2022. Retrieved 8 Jan 2022.
- ^ Szkoda, M.; Trzciński, Thousand.; Trykowski, G.; Łapiński, M.; Lisowska-Oleksiak, A. (5 December 2021). "Influence of brine metallic cations on the photoactivity of crystalline and exfoliated amorphous WO3 – photointercalation phenomenon". Practical Catalysis B: Environmental. 298: 120527. doi:10.1016/j.apcatb.2021.120527. ISSN 0926-3373. Archived from the original on 8 January 2022. Retrieved 8 Jan 2022.
- ^ Greenwood and Earnshaw, p. 84
- ^ Lincoln, S. F.; Richens, D. T.; Sykes, A. G. (2004). "Metal Aqua Ions". Comprehensive Coordination Chemical science II. p. 515. doi:10.1016/B0-08-043748-half-dozen/01055-0. ISBN978-0-08-043748-4.
- ^ Dean, John Aurie; Lange, Norbert Adolph (1998). Lange'south Handbook of Chemistry. McGraw-Colina. ISBN978-0-07-016384-three.
- ^ Burgess, J. (1978). Metal Ions in Solution. New York: Ellis Horwood. ISBN978-0-85312-027-viii.
- ^ Starks, Charles 1000.; Liotta, Charles L.; Halpern, Marc (1994). Stage-Transfer Catalysis: Fundamentals, Applications, and Industrial Perspectives. Chapman & Hall. p. 162. ISBN978-0-412-04071-9. OCLC 28027599.
- ^ Levy, One thousand. B. (1981). "Determination of Sodium with Ion-Selective Electrodes". Clinical Chemistry. 27 (viii): 1435–1438. doi:10.1093/clinchem/27.eight.1435. PMID 7273405. Archived from the original on 5 February 2016. Retrieved 26 November 2011.
- ^ Ivor L. Simmons, ed. (6 Dec 2012). Applications of the Newer Techniques of Analysis. Springer Science & Business Media, 2012. p. 160. ISBN978-1-4684-3318-0.
- ^ Xu Hou, ed. (22 June 2016). Pattern, Fabrication, Backdrop and Applications of Smart and Avant-garde Materials (illustrated ed.). CRC Printing, 2016. p. 175. ISBN978-1-4987-2249-0.
- ^ Nikos Hadjichristidis; Akira Hirao, eds. (2015). Anionic Polymerization: Principles, Practice, Forcefulness, Consequences and Applications (illustrated ed.). Springer. p. 349. ISBN978-4-431-54186-8.
- ^ Dye, J. L.; Ceraso, J. M.; Mei Lok Tak; Barnett, B. L.; Tehan, F. J. (1974). "Crystalline Common salt of the Sodium Anion (Na−)". J. Am. Chem. Soc. 96 (ii): 608–609. doi:x.1021/ja00809a060.
- ^ Holleman, A. F.; Wiberg, East.; Wiberg, N. (2001). Inorganic Chemistry. Academic Press. ISBN978-0-12-352651-9. OCLC 48056955.
- ^ Renfrow, W. B. Jr.; Hauser, C. R. (1943). "Triphenylmethylsodium". Organic Syntheses. ; Collective Book, vol. 2, p. 607
- ^ Greenwood and Earnshaw, p. 111
- ^ a b Paul Ashworth; Janet Chetland (31 December 1991). Brian, Pearson (ed.). Speciality chemicals: Innovations in industrial synthesis and applications (illustrated ed.). London: Elsevier Technology. pp. 259–278. ISBN978-1-85166-646-ane. Archived from the original on 16 December 2021. Retrieved 27 July 2021.
- ^ Habashi, Fathi (21 Nov 2008). Alloys: Grooming, Backdrop, Applications. John Wiley & Sons, 2008. pp. 278–280. ISBN978-3-527-61192-viii.
- ^ a b Newton, David E. (1999). Baker, Lawrence Due west. (ed.). Chemical Elements. U·X·Fifty. ISBN978-0-7876-2847-five. OCLC 39778687.
- ^ Davy, Humphry (1808). "On some new phenomena of chemical changes produced by electricity, specially the decomposition of the fixed alkalies, and the exhibition of the new substances which constitute their bases; and on the general nature of alkaline bodies". Philosophical Transactions of the Majestic Society of London. 98: i–44. doi:10.1098/rstl.1808.0001. Archived from the original on 12 March 2021. Retrieved v April 2021.
- ^ Weeks, Mary Elvira (1932). "The discovery of the elements. 9. Three alkali metals: Potassium, sodium, and lithium". Journal of Chemical Pedagogy. 9 (6): 1035. Bibcode:1932JChEd...ix.1035W. doi:10.1021/ed009p1035.
- ^ Humphry Davy (1809) "Ueber einige neue Erscheinungen chemischer Veränderungen, welche durch dice Electricität bewirkt werden; insbesondere über die Zersetzung der feuerbeständigen Alkalien, die Darstellung der neuen Körper, welche ihre Basen ausmachen, und dice Natur der Alkalien überhaupt" (On some new phenomena of chemical changes that are accomplished by electricity; specially the decomposition of flame-resistant alkalis [i.due east., alkalies that cannot be reduced to their base metals past flames], the preparation of new substances that constitute their [metallic] bases, and the nature of alkalies generally), Annalen der Physik, 31 (2) : 113–175 ; see footnote p. 157. Archived vii Dec 2016 at the Wayback Car From p. 157: "In unserer deutschen Nomenclatur würde ich die Namen Kalium und Natronium vorschlagen, wenn human being nicht lieber bei den von Herrn Erman gebrauchten und von mehreren angenommenen Benennungen Kali-Metalloid and Natron-Metalloid, bis zur völligen Aufklärung der chemischen Natur dieser räthzelhaften Körper bleiben will. Oder vielleicht findet human being es noch zweckmässiger fürs Erste zwei Klassen zu machen, Metalle und Metalloide, und in dice letztere Kalium und Natronium zu setzen. — Gilbert." (In our German nomenclature, I would suggest the names Kalium and Natronium, if one would not rather go along with the appellations Kali-metalloid and Natron-metalloid which are used by Mr. Erman and accepted past several [people], until the consummate clarification of the chemical nature of these puzzling substances. Or perhaps one finds it yet more advisable for the present to create two classes, metals and metalloids, and to place Kalium and Natronium in the latter – Gilbert.)
- ^ J. Jacob Berzelius, Försök, att, genom användandet af den electrokemiska theorien och de kemiska proportionerna, grundlägga ett hire vettenskapligt organization för mineralogien [Attempt, by the use of electrochemical theory and chemical proportions, to found a pure scientific organization for mineralogy] (Stockholm, Sweden: A. Gadelius, 1814), p. 87.
- ^ van der Krogt, Peter. "Elementymology & Elements Multidict". Archived from the original on 23 January 2010. Retrieved eight June 2007.
- ^ Shortland, Andrew; Schachner, Lukas; Freestone, Ian; Tite, Michael (2006). "Natron as a flux in the early vitreous materials industry: sources, ancestry and reasons for pass up". Journal of Archaeological Science. 33 (4): 521–530. doi:ten.1016/j.jas.2005.09.011.
- ^ Kirchhoff, G.; Bunsen, R. (1860). "Chemische Analyse durch Spectralbeobachtungen" (PDF). Annalen der Physik und Chemie. 186 (half dozen): 161–189. Bibcode:1860AnP...186..161K. doi:10.1002/andp.18601860602. Archived (PDF) from the original on two March 2016. Retrieved thirty June 2019.
- ^ Greenwood and Earnshaw, p. 69.
- ^ Lide, David R. (xix June 2003). CRC Handbook of Chemistry and Physics, 84th Edition. CRC Handbook. CRC Printing. xiv: Abundance of Elements in the Earth's Crust and in the Sea. ISBN978-0-8493-0484-2. Archived from the original on 7 Dec 2016. Retrieved 3 July 2016.
- ^ "D-lines". Encyclopedia Britannica. spectroscopy. Archived from the original on seven Nov 2017. Retrieved 6 November 2017.
- ^ Welty, Daniel E.; Hobbs, L. M.; Kulkarni, Varsha P. (1994). "A high-resolution survey of interstellar Na I D1 lines". The Astrophysical Journal. 436: 152. Bibcode:1994ApJ...436..152W. doi:10.1086/174889.
- ^ "Mercury". NASA Solar Organization Exploration. In Depth. Archived from the original on 16 March 2020. Retrieved 29 Feb 2020.
- ^ Colaprete, A.; Sarantos, K.; Wooden, D. H.; Stubbs, T. J.; Melt, A. Yard.; Shirley, Thou. (2015). "How surface composition and meteoroid impacts mediate sodium and potassium in the lunar exosphere". Science. 351 (6270): 249–252. Bibcode:2016Sci...351..249C. doi:10.1126/scientific discipline.aad2380. PMID 26678876.
- ^ "Cometary neutral tail". astronomy.swin.edu.au. Cosmos. Archived from the original on 22 April 2018. Retrieved six November 2017.
- ^ Cremonese, One thousand.; Boehnhardt, H.; Crovisier, J.; Rauer, H.; Fitzsimmons, A.; Fulle, M.; et al. (1997). "Neutral sodium from Comet Hale–Bopp: A third blazon of tail". The Astrophysical Journal Letters. 490 (ii): L199–L202. arXiv:astro-ph/9710022. Bibcode:1997ApJ...490L.199C. doi:10.1086/311040. S2CID 119405749.
- ^ Redfield, Seth; Endl, Michael; Cochran, William D.; Koesterke, Lars (2008). "Sodium absorption from the exoplanetary temper of HD 189733b detected in the optical manual spectrum". The Astrophysical Journal. 673 (1): L87–L90. arXiv:0712.0761. Bibcode:2008ApJ...673L..87R. doi:x.1086/527475. S2CID 2028887.
- ^ a b Eggeman, Tim; Updated By Staff (2007). "Sodium and Sodium Alloys". Kirk-Othmer Encyclopedia of Chemical Technology. John Wiley & Sons. doi:ten.1002/0471238961.1915040912051311.a01.pub3. ISBN978-0-471-23896-half dozen.
- ^ Oesper, R. E.; Lemay, P. (1950). "Henri Sainte-Claire Deville, 1818–1881". Chymia. 3: 205–221. doi:10.2307/27757153. JSTOR 27757153.
- ^ Banks, Alton (1990). "Sodium". Journal of Chemical Education. 67 (12): 1046. Bibcode:1990JChEd..67.1046B. doi:x.1021/ed067p1046.
- ^ Pauling, Linus, General Chemical science, 1970 ed., Dover Publications
- ^ "Los Alamos National Laboratory – Sodium". Archived from the original on 3 May 2019. Retrieved 8 June 2007.
- ^ Sodium production. Royal Society Of Chemistry. 12 November 2012. Archived from the original on 11 December 2021. Retrieved 27 July 2021.
- ^ Sodium Metallic from French republic. DIANE Publishing. ISBN978-1-4578-1780-9.
- ^ Mark Anthony Benvenuto (24 February 2015). Industrial Chemical science: For Advanced Students (illustrated ed.). Walter de Gruyter GmbH & Co KG, 2015. ISBN978-3-11-038339-3.
- ^ Stanley Nusim, ed. (xix Apr 2016). Active Pharmaceutical Ingredients: Development, Manufacturing, and Regulation, Second Edition (2, illustrated, revised ed.). CRC Press, 2016. p. 303. ISBN978-1-4398-0339-4.
- ^ Remington, Joseph P. (2006). Beringer, Paul (ed.). Remington: The Science and Practice of Chemist's shop (21st ed.). Lippincott Williams & Wilkins. pp. 365–366. ISBN978-0-7817-4673-i. OCLC 60679584.
- ^ Paterova, Iva; Reitmaierova, Lenka; Cerveny, Libor (2022). "Rosaphen synthesis: Homogeneously catalyzed aldol condensation of cinnamaldehyde or hydrocinnamaldehyde with propanal followed by hydrogenation over ruthenium and nickel supported catalysts". Reaction Kinetics, Mechanisms and Catalysis. 135: 71–82. doi:10.1007/s11144-021-02123-4. S2CID 245709591. Archived from the original on eight January 2022. Retrieved 8 Jan 2022.
- ^ Wiberg, Egon; Wiberg, Nils; Holleman, A. F. (2001). Inorganic Chemistry. Bookish Press. pp. 1103–1104. ISBN978-0-12-352651-9. OCLC 48056955. Archived from the original on i February 2016. Retrieved 8 January 2016.
- ^ Harris, Jay C. (1949). Metal cleaning: bibliographical abstracts, 1842–1951. American Gild for Testing and Materials. p. 76. OCLC 1848092. Archived from the original on 18 May 2016. Retrieved 8 Jan 2016.
- ^ Lindsey, Jack Fifty. (1997). Practical illumination engineering. Fairmont Press. pp. 112–114. ISBN978-0-88173-212-two. OCLC 22184876. Archived from the original on 17 June 2016. Retrieved 8 January 2016.
- ^ Lerner, Leonid (16 Feb 2011). Modest-Scale Synthesis of Laboratory Reagents with Reaction Modeling. CRC Press. pp. 91–92. ISBN978-1-4398-1312-6. OCLC 669160695. Archived from the original on 12 May 2016. Retrieved 8 January 2016.
- ^ Sethi, Arun (1 January 2006). Systematic Laboratory Experiments in Organic Chemical science. New Age International. pp. 32–35. ISBN978-81-224-1491-ii. OCLC 86068991. Archived from the original on 29 April 2016. Retrieved 8 January 2016.
- ^ Smith, Michael (12 July 2011). Organic Synthesis (iii ed.). Academic Press, 2011. p. 455. ISBN978-0-12-415884-ix.
- ^ Solomons; Fryhle (2006). Organic Chemical science (8 ed.). John Wiley & Sons, 2006. p. 272. ISBN978-81-265-1050-4.
- ^ "Light amplification by stimulated emission of radiation Development for Sodium Light amplification by stimulated emission of radiation Guide Stars at ESO" (PDF). Domenico Bonaccini Calia, Yan Feng, Wolfgang Hackenberg, Ronald Holzlöhner, Luke Taylor, Steffan Lewis. Archived (PDF) from the original on 13 March 2016. Retrieved 11 September 2016.
- ^ van Rossen, Chiliad. L. C. Grand.; van Bleiswijk, H. (1912). "Über das Zustandsdiagramm der Kalium-Natriumlegierungen". Zeitschrift für Anorganische Chemie. 74: 152–156. doi:10.1002/zaac.19120740115. Archived from the original on 11 March 2020. Retrieved 30 June 2019.
- ^ Sodium as a Fast Reactor Coolant Archived 13 Jan 2013 at the Wayback Machine presented by Thomas H. Fanning. Nuclear Engineering Partitioning. U.S. Department of Energy. U.S. Nuclear Regulatory Commission. Topical Seminar Series on Sodium Fast Reactors. 3 May 2007
- ^ a b "Sodium-cooled Fast Reactor (SFR)" (PDF). Office of Nuclear Free energy, U.S. Department of Energy. eighteen February 2015. Archived (PDF) from the original on 10 January 2019. Retrieved 25 June 2017.
- ^ Fire and Explosion Hazards. Inquiry Publishing Service, 2011. 2011. p. 363. ISBN978-981-08-7724-eight.
- ^ Pavel Solomonovich Knopov; Panos K. Pardalos, eds. (2009). Simulation and Optimization Methods in Take a chance and Reliability Theory. Nova Science Publishers, 2009. p. 150. ISBN978-1-60456-658-1.
- ^ McKillop, Allan A. (1976). Proceedings of the Estrus Transfer and Fluid Mechanics Found. Stanford University Press, 1976. p. 97. ISBN978-0-8047-0917-0.
- ^ U.S. Diminutive Energy Commission. Reactor Handbook: Engineering (2 ed.). Interscience Publishers. p. 325.
- ^ A US US2949907 A, Tauschek Max J, "Coolant-filled poppet valve and method of making same", published 23 August 1960
- ^ "Sodium" (PDF). Northwestern University. Archived from the original (PDF) on 23 August 2011. Retrieved 21 November 2011.
- ^ "Sodium and Potassium Quick Wellness Facts". health.ltgovernors.com. Archived from the original on 30 June 2018. Retrieved seven November 2011.
- ^ "Sodium in diet". MedlinePlus, US National Library of Medicine. 5 October 2016. Archived from the original on 29 March 2019. Retrieved 23 July 2016.
- ^ "Reference Values for Elements". Dietary Reference Intakes Tables. Health Canada. 20 July 2005. Archived from the original on 29 May 2017. Retrieved 25 August 2016.
- ^ U.S. Department of Agriculture; U.Southward. Department of Wellness and Human Services (December 2010). Dietary Guidelines for Americans, 2010 (PDF) (7th ed.). p. 22. ISBN978-0-16-087941-viii. OCLC 738512922. Archived from the original (PDF) on 6 Feb 2011. Retrieved 23 November 2011.
- ^ "How much sodium should I eat per day?". American Center Association. 2016. Archived from the original on 28 September 2016. Retrieved 15 October 2016.
- ^ a b Patel, Yash; Joseph, Jacob (13 December 2020). "Sodium Intake and Heart Failure". International Journal of Molecular Sciences. 21 (24): 9474. doi:10.3390/ijms21249474. ISSN 1422-0067. PMC7763082. PMID 33322108.
- ^ CDC (28 February 2018). "The links between sodium, potassium, and your claret pressure". Centers for Disease Control and Prevention. Archived from the original on 17 January 2021. Retrieved v January 2021.
- ^ a b Geleijnse, J. K.; Kok, F. J.; Grobbee, D. E. (2004). "Impact of dietary and lifestyle factors on the prevalence of hypertension in Western populations". European Journal of Public Health. 14 (3): 235–239. doi:x.1093/eurpub/14.3.235. PMID 15369026.
- ^ Lawes, C. M.; Vander Hoorn, S.; Rodgers, A.; International Guild of Hypertension (2008). "Global brunt of blood-pressure-related disease, 2001" (PDF). Lancet. 371 (9623): 1513–1518. CiteSeerX10.1.ane.463.887. doi:10.1016/S0140-6736(08)60655-eight. PMID 18456100. S2CID 19315480. Archived from the original (PDF) on 26 October 2015. Retrieved 25 October 2017.
- ^ Armstrong, James (2011). General, Organic, and Biochemistry: An Practical Approach. Cengage Learning. pp. 48–. ISBN978-ane-133-16826-3.
- ^ Table Salt Conversion Archived 23 September 2014 at the Wayback Automobile. Traditionaloven.com. Retrieved on 11 November 2015.
- ^ a b "Use the Nutrition Facts Label to Reduce Your Intake of Sodium in Your Nutrition". US Food and Drug Administration. three January 2018. Archived from the original on 25 Jan 2018. Retrieved 2 February 2018.
- ^ a b Andrew Mente; et al. (2016). "Associations of urinary sodium excretion with cardiovascular events in individuals with and without hypertension: a pooled analysis of information from four studies". The Lancet. 388 (10043): 465–75. doi:ten.1016/S0140-6736(16)30467-vi. hdl:10379/16625. PMID 27216139. S2CID 44581906.
- ^ McGuire, Michelle; Beerman, Kathy A. (2011). Nutritional Sciences: From Fundamentals to Nutrient . Cengage Learning. p. 546. ISBN978-0-324-59864-three. OCLC 472704484.
- ^ Campbell, Neil (1987). Biology. Benjamin/Cummings. p. 795. ISBN978-0-8053-1840-i.
- ^ Srilakshmi, B. (2006). Nutrition Scientific discipline (second ed.). New Historic period International. p. 318. ISBN978-81-224-1633-vi. OCLC 173807260. Archived from the original on i February 2016. Retrieved 8 Jan 2016.
- ^ Pohl, Hanna R.; Wheeler, John Due south.; Murray, H. Edward (2013). "Sodium and Potassium in Health and Disease". In Astrid Sigel; Helmut Sigel; Roland K. O. Sigel (eds.). Interrelations between Essential Metallic Ions and Human Diseases. Metal Ions in Life Sciences. Vol. 13. Springer. pp. 29–47. doi:10.1007/978-94-007-7500-8_2. ISBN978-94-007-7499-5. PMID 24470088.
- ^ Kering, M. K. (2008). "Manganese Diet and Photosynthesis in NAD-malic enzyme C4 plants PhD dissertation" (PDF). University of Missouri-Columbia. Archived (PDF) from the original on 25 April 2012. Retrieved 9 November 2011.
- ^ Subbarao, M. V.; Ito, O.; Berry, West. L.; Wheeler, R. M. (2003). "Sodium—A Functional Plant Nutrient". Critical Reviews in Found Sciences. 22 (five): 391–416. doi:ten.1080/07352680390243495. S2CID 85111284.
- ^ Zhu, J. Grand. (2001). "Plant salt tolerance". Trends in Constitute Science. six (2): 66–71. doi:10.1016/S1360-1385(00)01838-0. PMID 11173290.
- ^ a b "Plants and salt ion toxicity". Plant Biological science. Archived from the original on 3 April 2012. Retrieved 2 November 2010.
- ^ "Sodium 262714". Sigma-Aldrich. Archived from the original on xv January 2016. Retrieved one October 2018.
- ^ Run a risk Rating Information for NFPA Fire Diamonds Archived 17 February 2015 at the Wayback Automobile. Ehs.neu.edu. Retrieved on 11 November 2015.
- ^ Angelici, R. J. (1999). Synthesis and Technique in Inorganic Chemistry. Mill Valley, CA: University Science Books. ISBN978-0-935702-48-4.
- ^ Routley, J. Gordon. Sodium Explosion Critically Burns Firefighters: Newton, Massachusetts. U. Southward. Burn Administration. FEMA, 2013.
- ^ a b c Prudent Practices in the Laboratory: Treatment and Disposal of Chemicals . National Inquiry Council (U.S.). Committee on Prudent Practices for Handling, Storage, and Disposal of Chemicals in Laboratories. National Academies, 1995. 1995. p. 390. ISBN9780309052290.
- ^ An, Deukkwang; Sunderland, Peter B.; Lathrop, Daniel P. (2013). "Suppression of sodium fires with liquid nitrogen" (PDF). Burn Safety Journal. 58: 204–207. doi:ten.1016/j.firesaf.2013.02.001. Archived from the original (PDF) on 8 August 2017.
- ^ Clough, W. S.; Garland, J. A. (i July 1970). Behaviour in the Temper of the Droplets from a Sodium Fire (Study). U.Southward. Department of Energy Office of Scientific and Technical Information. OSTI 4039364.
- ^ Ladwig, Thomas H. (1991). Industrial burn down prevention and protection. Van Nostrand Reinhold, 1991. p. 178. ISBN978-0-442-23678-half dozen.
- ^ a b Günter Kessler (8 May 2012). Sustainable and Safe Nuclear Fission Energy: Technology and Prophylactic of Fast and Thermal Nuclear Reactors (illustrated ed.). Springer Science & Business Media, 2012. p. 446. ISBN978-three-642-11990-3.
- ^ Gordon, Routley J. (25 October 1993). Sodium explosion critically burns firefighters, Newton, Massachusetts (Technical study). United States Burn down Administration. 75.
{{cite techreport}}: CS1 maint: date and yr (link)
Bibliography
- Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN978-0-08-037941-8.
External links
![]()
Wikiquote has quotations related to Sodium .
- Sodium at The Periodic Table of Videos (Academy of Nottingham)
- Etymology of "natrium" – source of symbol Na
- The Wooden Periodic Table Table'south Entry on Sodium
- Sodium isotopes data from The Berkeley Laboratory Isotopes Project's
Sodium Hydroxide Density G Ml,
Source: https://en.wikipedia.org/wiki/Sodium
Posted by: moorehicave.blogspot.com




0 Response to "Sodium Hydroxide Density G Ml"
Post a Comment